
A recent study of men with early-stage prostate cancer found no difference in 10-year death rates, regardless of whether their doctors actively monitored the cancers for signs of growth or eradicated the men’s cancers with surgery or radiation.
What does this study mean for patients? Based on research we have conducted on prostate cancer decision-making, the implications are clear: Patients need to find physicians who will interact with them the way a good financial counselor would, taking the time to understand them well enough to help them find the treatment that fits their goals.
Imagine a couple in their 40s who ask a financial counselor for advice on retirement planning, and the counselor tells them how much to invest in domestic and foreign stocks versus bonds versus real estate without asking them about their goals. A good counselor would find out what ages the couple wishes to retire at, what kind of retirement income they hope to live off of, how much risk they are willing to take to achieve their goals, and how devastated they would be if their high return investments go south, forcing them to delay retirement or reduce their retirement spending.
Far too often in medical care, physicians don’t behave like good financial counselors–they give treatment recommendations without taking the time to understand their patients’ goals. Consider early-stage prostate cancer, a typically slow-growing tumor that is not fatal for the vast majority of patients who receive the diagnosis. In some men, the tumor lies indolent for decades.
For that reason, men sometimes choose to monitor their cancers–have their doctors conduct regular blood tests or biopsies to see if the tumor is beginning to spread. Such monitoring has the advantage of being relatively noninvasive, but it can create anxiety for patients who wonder, every six months, whether their next checkup will bring bad news.
For that reason, some men prefer active treatments like surgery or radiation that eradicate their cancers and therefore reduce cancer-related anxiety. But these more active treatments have their own downsides–each treatment is relatively arduous, and they can cause both erectile dysfunction and urinary incontinence.
The choice between active treatment and active monitoring depends on a patient’s goals–on how they view the trade-off between outcomes like cancer-related anxiety and erectile dysfunction. When counseling patients with early-stage prostate cancer, physicians need to help patients focus on these trade-offs.
(To read the rest of this article, please visit Forbes.)
Doctors Can't Be Trusted to Tell Patients Whether They Should Receive Robotic Surgery
Patients often rely on physicians for information about their treatment alternatives. Unfortunately, that information is not always objective.
Consider a man with early stage prostate cancer interested in surgical removal of his tumor, but uncertain whether it is better for the surgery to be performed with the help of robotic technology. He asks his surgeon for advice, and the surgeon explains that, while robotic surgeries have some advantages (smaller incisions, less blood loss), the advantages are “tiny and unimportant.” And besides: “You do have some smaller incisions with the robotic, but if you added up all the incisions from all the ports and from the incision to remove the prostate itself, it ends up equaling about the same incision length.”
Can that physician’s description of robotic technology be trusted?
My friend and colleague Angie Fagerlin led a study of prostate cancer decision-making that took place across four Veterans Affairs medical centers. As part of that study, she audio-recorded clinical interactions between patients and their surgeons. In some of these interactions, patients asked about the pros and cons of robotic surgery.
As it turns out, some urologists worked at VAs that had access to robotic equipment and others did not. In an analysis lead by Karen Scherr (an MD/PhD student at Duke), our research team discovered that surgeons who had access to the robot were more generous in describing its advantages over standard “open” surgery. When no robot was available, physicians downplayed its advantages. This is illustrated in the following figure, showing the percent of statements physicians made indicating that robotic surgery was inferior, equivalent or superior to traditional surgical approaches:
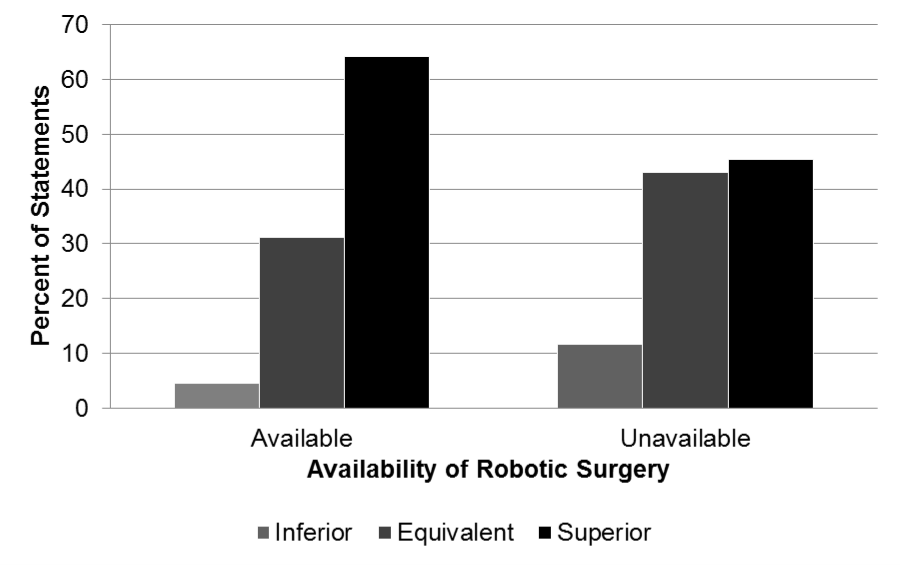
Importantly, I do not think physicians are willfully misleading patients about the pros and cons of robotic surgery. Instead, I think most physicians believe the robot is better (in the right hands) but when it is unavailable, doctors try to reassure patients that they will still receive state-of-the-art care. For example, one patient expressed anger that the robot was not available at his VA Hospital: “you see, that’s what’s so stupid about the VA!” The physician tried to assuage his concerns: “If you look at long-term outcomes related to cancer and cancer recurrence,” the surgeon said, “there has really been no difference. That’s why the VA system has not really invested in the robot.”
To read the rest of this article, please visit Forbes.
More Coverage of Our Out-Of-Pocket Expenses Research
My colleagues and I have been doing lots of research lately on how physicians and patients discuss out-of-pocket expenses during clinic encounters. One of our recent publications has been getting lots of attention, with this being the latest example. I thought I would share it with you:
Recent qualitative findings published in Health Affairs showed that physicians struggled to help patients factor out-of-pocket expenses into their medical decisions.
“Health care consumers cannot expect to make savvy financial decisions if their doctors do not engage with them in productive conversations about the pros and cons of their health care alternatives, including the financial costs,”Peter A. Ubel, MD, professor in the Sanford School of Public Policy at Duke University, told Healio Internal Medicine. “Some physicians say they are reluctant to do so because money talk would contaminate the doctor-patient relationship.”
In 2014, one in three Americans was reported to have difficulty paying health care bills, the researchers wrote. In a previous study, Ubel and colleagues found that patients and physicians discussed strategies to reduce health care expenses about 44% of the time when these topics arose. They also determined, however, that these discussions did not always allow patients to navigate out-of-pocket expenses.
“Many more physicians, I expect, would like to hold such conversations, but struggle to do so because there is no easy way to figure out how much patients will be required to pay out-of-pocket for their medical care,” Ubel said.
In the current study he and his colleagues performed a qualitative study by analyzing physician-patient interactions taken from the Verilogue Point-of-Practice database. They used 677 interactions in breast oncology, 656 in rheumatoid arthritis and 422 in psychiatry that occurred from May 2010 to February 2014 at private practices nationwide.
The researchers found that two broad categories of behaviors led to missed opportunities that could trim out-of-pocket expenses. The first was the physician not acknowledging the seriousness of the patient’s concerns. The second was the physician failing to fully resolve a patient’s financial concerns.
For the first behavior, the researchers cited examples of how physicians missed opportunities to address patient concerns.
To read the rest of this story, please visit Healio.
Thoughts on Shared Decision Making

I recently gave a talk about shared decision making at the annual conference for the National Comprehensive Cancer Network. Here is a nice write-up of that talk. For those of you silly enough not to travel to Florida to hear me pontificate!
After listening to the treatment alternatives—surveillance, or active treatment with surgery and radiation—a patient just diagnosed with prostate cancer asks you, “what would you recommend?”
How would you respond?
As a clinician, your responsibility is to find out what the patient is actually asking, and to provide a recommendation that reflects his or her preferences, Peter A. Ubel, MD, told attendees at the NCCN 21st Annual Conference.
One answer might be “I might make a different choice than you because I might have different preferences,” said Dr Ubel, the Madge and Dennis T. McLawhorn University Professor of Business, Public Policy, and Medicine at Duke University in Durham, NC.
Such a reply “is what should trigger the true shared decision-making conversation,” he added. Far too often, however, patients are inundated with information they either do not understand or find overwhelming in its sheer volume.
The “right” choice, he outlined, depends on how the patient weighs the trade-off between the anxiety of living with cancer and the side effects of the active treatments.
“To really empower patients to be partners in decisions where their preferences are important in determining what the right course of action is, we have to communicate better,” he said. “We have to understand decision psychology better than we do, and we have to make sure we know the difference between a medical fact and a value judgment.”
Dr Ubel, with the Duke-Margolis Center for Health Policy, provided an example of an urologist explaining a Gleason score to a patient: “low risk is Gleason 6, intermediate is usually 7s, with 3 + 4 or 4 + 3, depending on how it looks under the microscope. And then 8, 9, and 10 are all high risk. So it’s in the middle. It was 3 + 3 and 3 + 4, so just enough of the atypical cells of the grade 4 to make it 3 + 4, which means you’re intermediate risk.”
Although this represents an earnest explanatory effort, what the clinician is trying to say is, “you do not have the kind of cancer that will kill you, maybe never, but certainly not in the next 10 or 15 years. We have months to decide what to do about this. We found it early enough and we can take care of this.”
“That’s what the patient needed to understand, and instead, what ends up, is the doctor kind of forgets that the patient doesn’t speak medicalese,” he said.
Informed consent also plays a role, in that clinicians may feel the need “to inform the heck out of patients with all the medical detail [they] believe is necessary to understand the decision, instead of a translation of that medical detail into terms a patient can understand.”
One challenge with shared decision-making is that thorough communication is “not always good communication,” and “good communication takes time, to acknowledge and deal with emotion, to absorb and reflect on information,” he said. This includes the ability to take the patient’s perspective into account, with recommendations often evolving.
Dr Ubel outlined 3 approaches he believed clinicians “can do better to promote shared decision-making.”
“First off, we need to use a simple technique called ‘teach back.’ When you give people information about their treatment alternatives, don’t assume they understand it because they are nodding,” he said. “Find out if they understand it by asking them to tell you in their own words.” For example, “‘Can you tell me what you think you understand the alternatives mean?’”
“You will learn so much in hearing patients translate back to you what you just said to them. I guarantee you, you do that a dozen times…and by the time you get to that 12th patient, your spiel—because we all have spiels to describe information—your spiel will probably be half as long as it used to be and twice as good because you’ll realize, early on, that patients are not hearing most of what you say.”
Secondly, even if patients say they do not want to be full partners in the decision and want to be told what to do, clinicians need to get to know patients well enough to help them make the right choices.
“When a patient asks what you would recommend,” try to find out something about the patient first. “That is critical to giving good advice.”
Finally, what’s needed are “recommendations that are based on an understanding of patient preferences,” he said.
Dr Ubel concluded by noting that “we also should be careful about how much information we give people. People can be harmed by receiving too much information. Too much information makes it hard to comprehend, regardless of whether there’s jargon. You give too much information loaded with jargon and understanding is going to be very poor”—thus making choices more difficult.
The Wrong Way To React When Terminally Ill Patients Cry

Just three weeks earlier, she had noticed something strange about one of her breasts. An irregular shape. Her daughter brought her to the doctor, and soon the patient, I’ll call her Amanda, was diagnosed with breast cancer, stage “to be determined.” In fact, she was now in an oncologist’s office, learning what tests she would receive to determine the extent of her tumor. And sitting between her and the doctor was a tape recorder, capturing their conversation.
A dozen minutes into the appointment, Amanda would break down crying. And the physician’s response, which I will lay out for you in a bit, is unfortunately not uncommon. When patients express negative emotion, many oncologists do not respond with empathy. As I’ll explain later, this is an enormous problem, but also one we can fix.
Amanda was 60 years old at the time of the appointment, quite frail for her age, requiring help climbing up onto the exam table because of a recent stroke. She needed to wear adult diapers. She also suffered from diabetes and tremors, although it was unclear whether those non-spontaneous movements were from Parkinson’s or some milder disorder. In other words, her health was already fragile and a breast cancer diagnosis wasn’t going to make things better.
Which may be why she was so distraught about her situation.
The oncologist described how he would evaluate her problem: “Now I am going to order a scan, a CT scan. It’s like an x-ray but she needs to lie down,” he explained to Amanda’s brother. “After that, we will check her blood. After we’ve done the blood test and the scan, I will meet you in one week and we will discuss this, and I will advise accordingly.”
Then, perhaps noticing the look on Amanda’s face, he advised her: “Don’t be scared, please. We will wait for the scan and blood results and see you in one week. So next week, [turning to the brother] please come with your children [one of whom was Amanda’s caregiver] and I can discuss this further.” Amanda’s brother agreed with the plan, but Amanda started crying: “So difficult,” she said.
Her brother tried to intervene. “Stop crying,” he said. The oncologist also stepped into the uncomfortable situation: “Amanda, don’t be scared, please. We don’t know for sure [how bad your cancer is], so let us check first. OK?”
“Doctor will do the best for you,” continued her brother, “so don’t cry. OK?” The physician continued, almost a tag team now with the brother. “Today we can do the blood test. You don’t have to wait after doing that and can go home thereafter.”
“You have a lot of work, right?” she said, apologizing for letting her emotions take up so much of the doctor’s time. He tried to ease her mind. “No,” said the doctor, denying that he was too busy to address her concerns. But he immediately muddled his message. “I mean, you can do your blood tests today.”
A heartbreaking episode, heartbreaking in large part because of the awful situation poor Amanda was in, with so many things she could no longer do because of health problems and now with advanced cancer. Tragic, truly tragic. But compounding this tragedy was a veritable tragicomedy of miscommunication. Amanda breaks down crying and what message does she hear from her brother and doctor?
Stop crying.
Neither brother nor doctor acknowledged that, given her situation, she had a right to be scared, that it would in fact be abnormal not to be frightened. Neither realized that when people start crying, telling them to “stop crying” can actually make patients feel worse. I am sure I have made this same mistake scores of times in my own clinical practice. When a patient cries, our natural instinct as doctors, as humans, is to relieve their suffering, to say something that will stop their crying. It is perfectly normal, even compassionate, to reach out to soothe someone who is crying, to gently tell them not to cry, that everything will be okay.
(To read the rest of this article, please visit Forbes.)
Has Mammography Created an Epidemic of Pseudo-Survivorship?

Karen Vogt’s breast cancer journey began like many others, with her breasts painfully squeezed into a mammography machine. At age 52, it was far from her first mammogram, but this scan would be the most consequential by far. It revealed microcalcifications, little areas of breast tissue speckled with deposits of calcium that her radiologist worried were suspicious for a nascent cancer, especially since these specks hadn’t been so conspicuous twelve months earlier. A biopsy proved that the radiologist’s suspicions were warranted. Vogt had a small cancer in her left breast, a ductal carcinoma in situ, as her doctors called it. Stage 0 cancer. What should she do?
This week, medical researchers published a study showing that when women are diagnosed with stage 0 breast cancer, no matter what treatment they receive, their life expectancy is equivalent to women who were never diagnosed with breast cancer. This finding further fuels debates about whether we are screening for and/or treating breast cancer too aggressively. Last year, in fact, a study came out suggesting that even in women 50 years of age and older, annual mammographies are not the life-savers that they were made out to be by medical experts. According to these studies, for every woman like Vogt with a cancer detected by mammography, hundreds more will go through the painful test without any cancer being detected and dozens will experience the harms of “false positive” test results, a term medical experts use to refer to abnormal findings which do not turn out to be a cancer. And just this week, a study was published showing that stage 0 breast cancers are better off untreated
Yet despite increasing evidence of the significant harms of mammography, compared to its relatively modest benefits, many American women dutifully continue to receive annual tests. Why do they remain enthusiastic about mammography? In large part because many women who were harmed by mammography believe the opposite. By identifying non-invasive lesions, like the DCIS discovered in Karen Vogt, mammography has created a community of women incorrectly convinced that the test saved their lives.
Has overuse of mammography created a false epidemic of breast cancer “survivorship?” (To read the rest of this article, please visit Forbes.)
New Review of Critical Decisions
My heart was warmed by a new, and very positive, review of a Critical Decisions. I always find myself agreeing with people who liked my book. Funny thing. Anyway, here is the review:
“This is a very belated review. And I mean that as a compliment.
As soon as I finished reading Peter Ubel’s Critical Decisions, I immediately leant it out to a colleague as a very enjoyable “must-read.” She devoured it and passed it along to yet another colleague – and that was literally the last I saw of my copy, which I hope to eventually get back in some kind of battered but well-read condition.
Yes, we’ve ordered more copies. But I think everyone keeps passing it on because Critical Decisions provides a wealth of history and information about medical decision making, and how doctors and patients each think about decisions. Perhaps most importantly, it shows the author confronting the reality of how medical decision making actually plays out in the real world.
It starts by tracing the history of medical decisions. Yes, we’ve come a long way from the days where even a U.S. president wouldn’t question a physician’s recommendation for his wife to get something as life-changing as a mastectomy without even discussing the biopsy results, to a world where individuals and families have more opportunity to participate in and make decisions about their care.
But on both the patient and the clinician side, so many factors influence and bias us, and we’re often unaware of it.
First, as Ubel points out, decisions are also a burden. And when decisions get even a little tough we often look for ways not to decide. Yep, we defer. A common instinct is to ask our doctor what they would do if they were the patient. But it’s a problematic request, because our judgment changes when we’re actually responsible for the decision.
Even if we don’t defer, we often base decisions on a friend or family member’s good or bad experience with a drug or treatment, which may or may not be the norm. We also underestimate our own resilience and can’t imagine getting used to things like self-injecting insulin. And when words like “cancer” are introduced, we inflate risks, thinking they’re more likely. Clearly, it’s hard to give our brains space for calm deliberation. And, as Ubel writes:
Good decision making is not merely a matter of comprehension. Understanding doesn’t necessarily lead people toward making rational choices. Decision making is often as much about feeling as it is about thinking. And the way people use risk information… is often influenced less by their understanding of the magnitude of these risks than by subtle psychological cues that change the way these risks feel. (p. 137)… (Read more)”
(Click here to view comments)
Discussing Financial Toxicity in Oncology Settings
 The American Society of Clinical Oncology is one of the leading specialty organizations for physicians who take care of patients with cancer. A reporter from ASCO just wrote a nice piece, in which she questioned me to go into more detail about the challenge of discussing cost of care with oncology patients. I thought I would share that with you:
The American Society of Clinical Oncology is one of the leading specialty organizations for physicians who take care of patients with cancer. A reporter from ASCO just wrote a nice piece, in which she questioned me to go into more detail about the challenge of discussing cost of care with oncology patients. I thought I would share that with you:
High costs of cancer treatments can be an “undisclosed toxicity” that can harm a patient’s overall health and well-being, according to an article in The New England Journal of Medicine. High medical bills can not only cause stress and anxiety but may also compel patients to cut back on spending for other basic needs—such as food, leading to less healthy diets—or to take medications less frequently than prescribed.
“This is a very frequent cause of nonadherence,” the article’s lead author,Peter A. Ubel, MD, Professor of Business, Public Policy, and Medicine at Duke University, Durham, North Carolina, said in an interview with The ASCO Post. “It is a medical problem. Patients may not be showing up for tests or taking their pills because they can’t afford it. Dr. Ubel also tackled the issue of physicians rarely discussing medical intervention costs in an op-ed article he wrote for The New York Times.
Not Always Easy to Know
“Because treatments can be ‘financially toxic,’ imposing out-of-pocket costs that may impair patients’ well-being, we contend that physicians need to disclose the financial consequences of treatment alternatives just as they inform patients about treatments’ side effects,” Dr. Ubel and colleagues wrote…(Read more here)
Getting Good Cancer Care by Asking the Right Questions

Not long ago, I spoke with a freelance journalist, Charlotte Huff, who put together a really nice article in Cancer Today, describing how cancer patients can get more engaged in their care. I thought I would share a bit of that piece with you today:
Gloria Full wasn’t surprised that the recommended chemotherapy regimen was aggressive. She and her oncologist were, as Full puts it, ‘running scared.’ She had completed treatment for stage IV diffuse large B-cell lymphoma in 2006. Five years later, in 2011, a PET scan and subsequent biopsy identified a mass in her nasal cavity. “First thing I thought was, ‘Oh, my lord. It’s too close to the brain, so let’s get going,’ ” she recalls.
Full, a 68-year-old retired social worker who lives in Phoenix, was started on the platinum-based chemotherapy combination dubbed DHAP (dexamethasone, cytarabine and cisplatin). Halfway through the aggressive regimen, she and her doctor worried that it was inflicting too much damage on her already suppressed immune system. He suggested a PET scan to see if the treatment had offered any benefit so far, and to determine what to do next. “On that first one, it showed that the mass had become smaller,” Full says. Armed with that good news, she agreed with her doctor’s recommendation to cease any further cycles of treatment… (Read more here)
Shared Decision Making in Medicine
 The urologist broke the news: “Out of 12 cores, three were positive for cancer, so you have a small amount of cancer.”
The urologist broke the news: “Out of 12 cores, three were positive for cancer, so you have a small amount of cancer.”
He would soon explain the treatment choices—surgery, radiation, or active surveillance (watching the cancer closely with blood tests and biopsies). He described each option in elaborate detail, because he knew that the “right choice” would depend on what his 70-year-old patient thought about the pros and cons of treatment versus surveillance.
But before explaining the choices, the urologist wanted to make sure that the man in front of him understood the limited extent of his cancer:
We grade prostate cancer on how it looks under the microscope. We give it a score between 6 and 10.
Is that the Teason? the patient asked.
That’s the Gleason score.
Oh, Gleason score, OK.
Yep, so 6 is what we consider the lowest grade, least aggressive-looking, but it’s just abnormal enough for us to call it cancer. If it were any less than that—if there were less atypical-looking cells—we couldn’t call it cancer. So it’s just enough to get a grade of cancer, and then that goes all the way up to a score of 10, which is very abnormal looking and is more aggressive.
But 6 is the beginning number?
Yes, 6 is the least aggressive, 10 is the most aggressive, the doctor clarified.
I’m used to like, 1, the patient said, laughing.
Yeah well, the way we typically split it up is into thirds; low risk, intermediate risk, and high risk.
Right, said the patient.
Low risk is Gleason 6, intermediate is usually 7’s—3+4 or 4+3, depending on how it looks under the microscope. And then 8, 9, and 10 are all high risk. Yours is an intermediate risk. So it’s in the middle. It is 3+3 and 3+4, so just enough of the atypical cells of the grade 4 to make it 3+4, which means you’re intermediate risk.
The conversation is remarkable not just for its inscrutability but for the very fact that it took place… (Read more here)

