On Health,
Bioethics, and
Behavioral Science
As a physician and behavioral scientist at Duke University, my research and writing explore the quirks in human nature that influence our lives — the mixture of rational and irrational forces that affect our health, our happiness, and the way our society functions.

Latest Publications
Debt by Design — Navigating the Hazards of Medical Credit Cards
Amid increases in both out-of-pocket health care costs and enrollment in high-deductible insurance plans in the United States, medical…
Who’s Deciding? A Study of Patient-Surgeon Discussions About Contralateral Prophylactic Mastectomy
In this study, we explore conversations about contralateral prophylactic mastectomy–about whether to remove a healthy breast when also removing…
When crowdfunding for medical expenses, who gets the money?
Led by a former Duke medical student, Steven Doerstling, we analyzed, oh, 90K online campaigns, where people (or their…
Health Care in an Evolving Immigration Landscape — Providing Care while Upholding the Law
Many clinicians and healthcare provider organizations are struggling to figure out how to do their jobs with all the…
Integrating Out-of-Pocket Costs Into Shared Decision-Making for Heart Failure With Reduced Ejection Fraction
Led by the amazing Neal Dickert, cardiologist at Emory, we conducted an RCT of an intervention designed to promote…
Physician Gender and Patient Perceptions of Interpersonal and Technical Skills in Online Reviews
Do patients’ on-line physician reviews reveal gender bias? In a study led by the marvelous Dr. Farrah Madanay, we…
Latest Blog Posts
Private Equity-Owned Retinal Practices Perform Fewer Retinal Detachment Procedures
In recent years, many private equity firms have bought up physician practices, and then looked for ways to increase…
Another Way Coffee Won’t Kill You
Coffee has been falsely accused of causing a host of health problems. However, one potential risk of coffee has…
Medicare Can Save $4 Billion On Four Cancer Drugs — Can You Guess Which Ones?
Medicare could save more than $4 billion by negotiating prices on just four drugs. Want to guess what those…
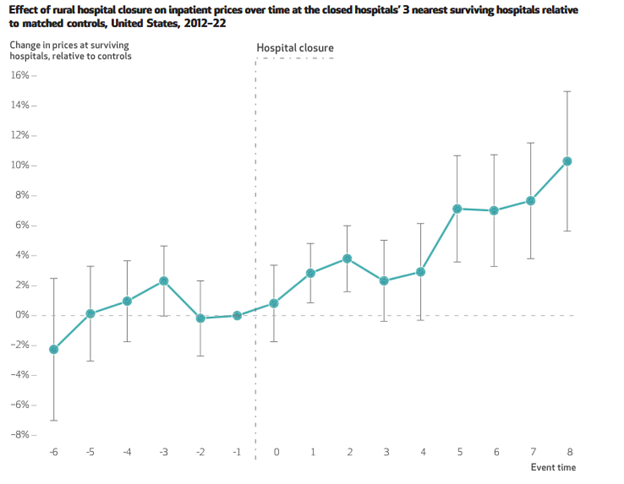
Upset About The High Price Of Your Hospital Stay? Medicaid Cuts Might Be To Blame
Under the Big Beautiful Bill, the federal government is making steep cuts to Medicaid funding. Those cuts could increase…
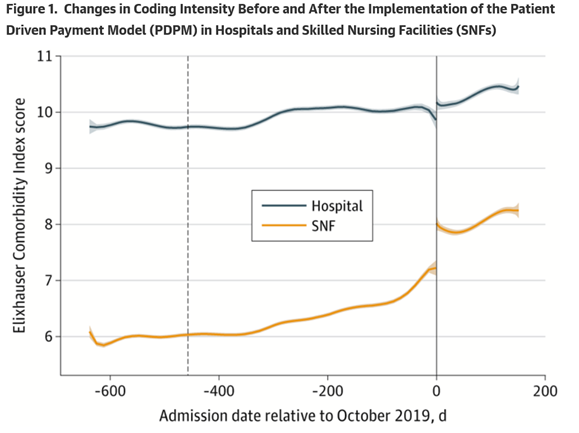
Are Nursing Homes Lying About Their Patients To Increase Profits? You Decide
Imagine you are CEO of a chain of nursing homes. You know that the sicker your patients are, the…
Thinking About IV Hydration At A Nearby Medical Spa? Think Again
On a hot summer day in Texas two years ago, Jenifer Cleveland visited a medical spa to receive IV…